Disease X was supposed to be caused by an influenza virus.
For years, scientists, world leaders, and other critical stakeholders have been preparing for the moment when a novel zoonotic pathogen would cross the species barrier from an animal population into the global human population in a big way. This is called a spillover event, and in role-playing exercises, the resulting hypothetical global pandemic came to be known as Disease X.
The last time a disease emerged on this scale was the 1918 influenza pandemic, and the last influenza spillover was the H1N1 “swine flu” in 2009. Influenza met all the criteria for a problem pathogen from a pandemic preparedness perspective: 1) it spreads rapidly from person-to-person, 2) it can hide and mutate in animal reservoirs (particularly birds and pigs), and 3) it can impact many parts of society, including the food supply. Because H1N1 could infect pigs, many animals were culled and the pork supply chain was badly affected.
COVID-19 is worse, and its impacts on the food supply are far greater than many imagined.

Like H1N1 influenza, SARS-CoV-2 (the virus that causes COVID-19 disease) infected people through a spillover event, most likely from a bat, although another animal may have been involved as well. At first, case trace back from the December outbreak of pneumonia in Wuhan, China implicated a seafood and “wet market,” a place where wildlife and other live animals are bought and sold. These are considered to be risky places from a pandemic preparedness perspective, since stressed wild animals from many different areas are brought together—a recipe to encourage a pathogen to jump species. However, later genetic work on SARS-CoV-2 and additional information suggested that the virus may actually have emerged as early as November, before the wet market outbreak. Currently, many questions remain about the origins of the virus, although scientists agree that it is zoonotic (comes from animals) and that a bat coronavirus is the closest relative to SARS-CoV-2. The figure below shows the origin and co-evolution of the coronavirus family prior to the SARS-CoV-2 emergence.
Given the potential for influenza to infect pigs, preparation for Disease X assumed that one impact to the food supply would be culling related to pigs, chickens or other livestock becoming infected, suggesting a need to limit the spread in the animal population through depopulation of affected farms.
With COVID-19, animals are being depopulated, but not because of animal infection (in fact, preliminary animal studies suggest pigs and chickens don’t easily get infected with SARS-CoV-2). Instead, they are being depopulated because of the impacts to the health of humans who work on farms and in meat processing plants, leading to plant closures. If the animals can’t be harvested, they can’t be kept indefinitely, consuming expensive grain and outgrowing the equipment that would be used to harvest them later. To complicate the situation, the feed supply chain, which is global, also has been disrupted by COVID-19 mitigation measures—the average person might run to the store to stock up on toilet paper, and livestock producers run to the futures market to stock up on grain.
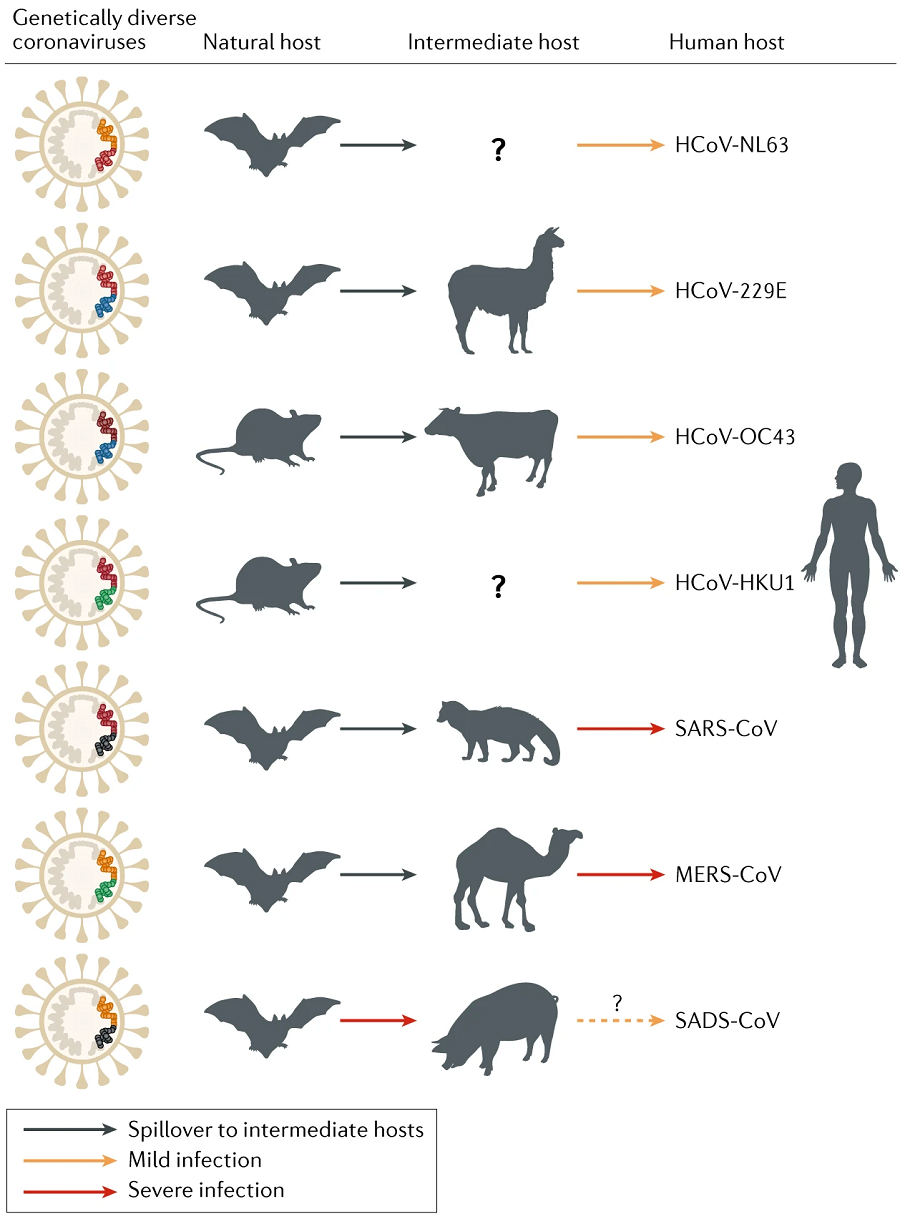
Source: Fang and Shi (2019)
So what to do to avoid a future Disease X? First, researchers and development practitioners must recognise that the health of people, animals and our shared environment are tightly interconnected. Public health issues are environmental issues, and taking a One Health approach to science, is critical to avoid future zoonotic spillovers. USAID’s PREDICT project encompasses transdisciplinary teams made up of public health specialists, epidemiologists, veterinarians, and environmentalists. These teams focus on geographic hotspots where wildlife is most likely to carry zoonotic diseases that could be a threat to human health and food systems. They catalogue diseases, create predictive maps of potential disease outbreaks and help communities respond to the threat of an outbreak.
Second, we need to understand that changes in ecosystems where animals live are driving the circulation of viral spread. Wildlife’s habitats are threatened and affected by land use changes and deforestation, often due to agriculture expansion to grow more food, ironically. Trade of wildlife is common and found for purchase in wet markets in many parts of the world. This is shifting viral reservoirs and contact-rates between virus carrying animals and humans. Regulation of illegal sales of wildlife in global food trade and food markets must be governed by local authorities, and balancing respect for cultural food practices with public health prevention measures will be important.
Third, social behaviour change strategies should be prioritised and scaled up that focus on issues of secondary infections and handling of animals. Examples of campaigns in Sierra Leone with Lassa fever and in the DRC with Ebola increased awareness of how disease is transmitted in the handling and exposure of animals as food or animals contaminating food.
And last, there is no systematic global effort to monitor pathogens emerging from animals that put human populations at risk. There is a need for even more surveillance through traceability technologies in the food system to track potential zoonotic and food-borne illnesses that are and will continue to present a threat to food systems and the health of our global population. One Health approaches—those at the intersection of human health, animal health, and environmental health—are critical as we respond to COVID-19, recover and learn from its impacts, and prepare for the next Disease X.
Meghan Davis is Associate Professor at Johns Hopkins Bloomberg School of Public Health in the Department of Environmental Health and Engineering. She is a molecular epidemiologist and an environmental microbiologist, focused on the interface of bacteria and hosts to reduce microbe-mediated disease in humans and animals. Her work applies the principles of one health and microbial ecology, evaluating target microbes and bacterial genes specifically and the larger microbial community (microbiome) broadly. Read more.
Jessica Fanzo is Bloomberg Distinguished Professor of Global Food & Agricultural Policy and Ethics at the Nitze School of Advanced International Studies (SAIS), the Berman Institute of Bioethics, and the Department of International Health of the Bloomberg School of Public Health at Johns Hopkins University. She also serves as the Director of the Global Food Ethics and Policy Program at Hopkins, and plays key advisory roles in Johns Hopkins’ Alliance for a Healthier World on the food security and nutrition theme, as well as the Bloomberg American Health Initiative on obesity and food systems. Read more.
